CRYO ARM™ 200 (JEM-Z200FSC)
Field Emission Cryo-Electron
Microscope

Cryo-electron microscopy has been established as a method to enable observation of cells and biological molecules with no fixation and no staining. Owing to the recent rapid progress of hardware and software, this microscopy technique has become increasingly important as an atomic-scale structural analysis method. In addition, technologies that enable analysis of membrane proteins without crystallization have been developed, resulting in increased use of cryo-electron microscopy for drug discovery. Thus, installation of cryo-electron microscopes (cryo-EM) in universities and research laboratories is greatly accelerating. To meet the needs of cryo-EM users, JEOL has developed a new cryo-EM "CRYO ARM™ 200", which automatically acquires image data for Single Particle Analysis over a long period of time.
Features
Automated specimen exchange system
The system is composed of a specimen stage to cool samples at liquid nitrogen temperature and a cryo-transfer system to automatically transfer the cooled samples to the cryo-stage. Liquid nitrogen is automatically supplied to the liquid nitrogen tank as required. This automated system features the storing of up to 12 samples and the exchange of arbitrary one or more samples while the rest of samples are kept cooled between the specimen stage and specimen exchange system. These unique capabilities enable flexible scheduling of microscopy.
Cold field emission gun (Cold FEG)
Cold FEG produces a high-brightness electron beam with very small energy spread, offering high coherency. Thus, the CRYO ARM™ 200 achieves high resolution, high contrast imaging.
Incolumn energy filter (omega filter)
Equipped with an incolumn energy filter (omega filter), the CRYO ARM™ 200 acquires energy filtered images and energy loss spectra. Zero-loss image acquired with the microscope provides high contrast with reduced chromatic aberration.
Automated image acquisition software for Single Particle Analysis
The CRYO ARM™ 200 incorporates automated software. The software allows for automated detection of holes on the specimen grid for efficient acquisition of Single Particle Analysis images.
Hole-free phase plate *1
This unique phase plate is suitable for higher contrast in biological specimens which originally provide only low contrast.
Auto adjustment functions *2
Auto focus, auto coma-free alignment, auto parallel-beam illumination and other automated adjustments are available, enabling image acquisition under optimum conditions.
Optional unit.
Images acquired with the bottom mount camera are used.
Specifications
Main instrument
| Electron gun | Cold field emission gun (Cold FEG) |
|---|---|
| Accelerating voltage | 200kV |
| Energy filter | In-column Omega energy filter |
| Maximum specimen tilt angle | ± 70° |
Schottky field emission gun can optionally be configured.
Specimen Stage / Automated specimen exchange system
| Specimen stage | |
|---|---|
| Coolant | Liquid nitrogen Automated liquid-nitrogen filling system built-in |
| Specimen cooling temperature | 100K or less |
| Temperature measurement position | Specimen, Cryo-shield, LN2 tank |
| Specimen movements | |
| X、Y | Motor drive (movements: ±1 mm) Piezoelectric elements (movements: ±0.5 μm) |
| Z | Motor drive (movements: ±0.2 mm) |
| Tilt-X | Motor drive (tilts: ±70°) |
| Rotation within the specimen plane | 0° or 90° |
| Specimen exchange system | Air-lock Automated cryo-transfer system built-in |
| Cooling temperature (specimen exchange chamber) |
105K or less |
| Specimen exchange cartridge | Up to 4 specimens can be changed at one time. |
| Specimen parking stage | Up to 12 specimens can be held. |
Catalogue Download
CRYO ARM™ 200 (JEM-Z200FSC) Field Emission Cryo-Electron
Application
Application JEM-Z200FSC
Exploring Biological Samples in 3D Beyond Classic Electron Tomography
Atomic-Resolution Elemental Mapping by EELS and XEDS in Aberration Corrected STEM
Gallery
Innexin-6 gap junction channel
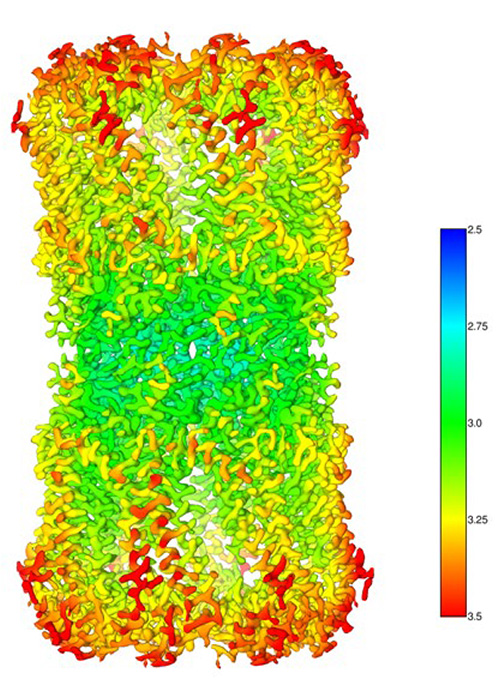
Experimental conditions
Sample: Innexin-6 (Caenorhabditis elegans)
Microscope: CRYO ARM™300 (300kV CFEG) with Gatan K2
Software used for image acquisition : JADAS (1974 images)
Number of particles: 91,613 (Initial pickup), 37,767 (for 3D reconstruction)
Software used for image analysis: Relion3
Resolution: 3.0 Å (at FSC = 0.143)
Sample by courtesy of Prof. A. Oshima (Nagoya University)
Apoferritin
The highest resolution 1.53 Å achieved by cryoEM 2019.02
mouse apoferritin


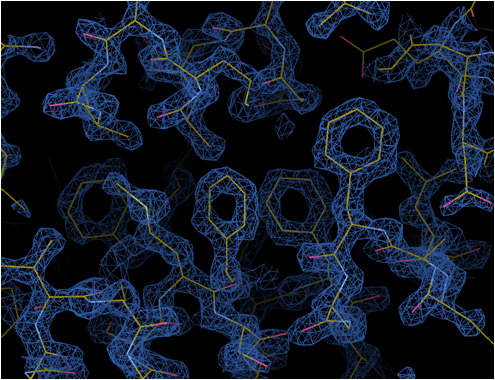
Phenylalanine

Methionine sulfur atom
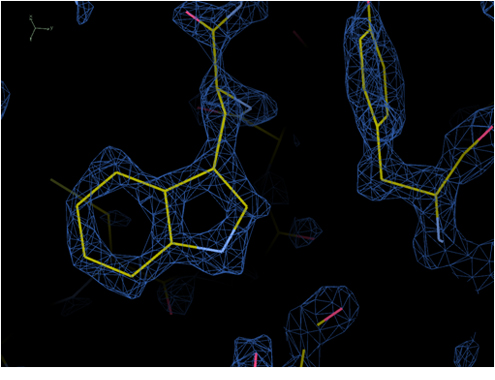
Tryptophan
Optics features: Cold FEG 300 kV & Ω-type energy filter with 20 eV slit width
Detector: Gatan K2 (image pixel size: 0.495 Å, mag x100,000)
Grid: Quantifoil 1.2/1.3 Cu 200 mesh, kept in the autoloader for 3 days before data collection
No. of micrographs: 974 collected over 24 h, 840 used for image analysis
No. of particles: 120,295 used for final reconstruction
Software: RELION 3.1b, CTFFIND4
Resolution: 1.53 Å (B-factor: 47)
Note: the first 56 images alone produced a map of 1.76 Å resolution (B-factor: 45)
Mouse apoferritin plasmid from Yanagisawa, Danev & Kikkawa @Tokyo University
Kato, Makino, Nakane, Terahara, Kaneko, Shimizu, Motoki, Ishikawa, Yonekura & Namba 2019.02 (EMDB-9865)
ß-galactosidase
β-galactosidase 2.43 Å resolution CRYO ARM™
β-galactosidase 2.43 Å resolution CRYO ARM™
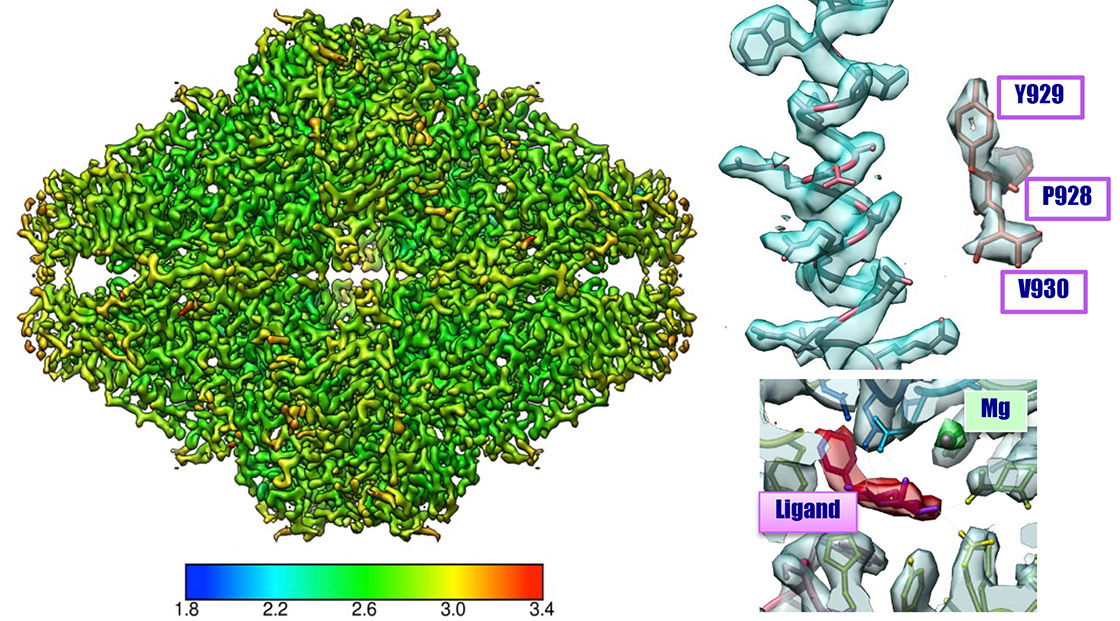
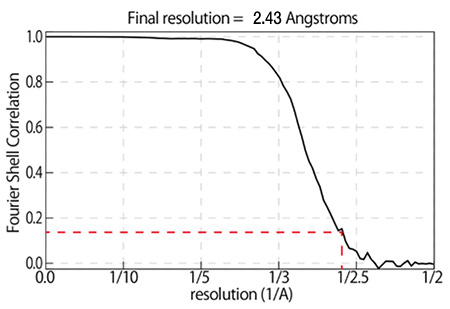
Sample:
β-galactosidase with PETGMicroscope:
CRYO ARM™ (Schottky 200 kV) / K2 summitNumber of Images:
2,500 over 3 days by JADASImage pixel size:
0.8 Å/pixelNumber of particle images:
350,000(Initial pickup), 88,564 (for final 3D reconstruction)Software:
Motioncor2, Gctf, Gautomatch, Relion2.0Total dose:
70 e-/Å2 (70 frames (0.2 sec/frames x 14 sec)
Data: courtesy by Dr. T. Kato and Dr. K. Namba, Osaka University, August 2017
Related Products

SightSKY Camera (EM-04500SKY) High sensitivity, Low noise fiber coupling CMOS camera
High-sensitivity, low-noise 19 M pixel CMOS sensor enables clearer imaging with fine specimen details observable even at low electron doses.
Its global shutter and high frame rate (58 fps/full pixel mode) enable image series acquisitions with less artifacts during dynamic observation.
"SightX" camera system control software provides user-friendly operations.
More Info
Are you a medical professional or personnel engaged in medical care?
No
Please be reminded that these pages are not intended to provide the general public with information about the products.
