Hydrogen Detection in Steel Pins by Direct-EGAMS
MSTips No. 412
Introduction
Recently, efforts are being made to create a decarbonized society as an issue of global environmental conservation, and it is necessary to strengthen the reduction of greenhouse gases such as carbon dioxide. In particular, material industries such as steel and chemicals use a large amount of coal and petroleum in their manufacturing processes, so they emit a large amount of CO2. Especially in the steelmaking process, the utilization of hydrogen is expected. On the other hand, hydrogen embrittlement of steel materials due to hydrogen absorbed in steel materials also exists as a problem, and analytical techniques for confirming the presence of hydrogen in steel materials are required. Therefore, we introduce an analysis example in which the hydrogen generated when steel pins are heated using a pyrolyzer is directly detected by a mass spectrometer.
Experimental
A commercially available steel pin (containing 7.9 ppm hydrogen) was used as the sample. For the measurement, a
system in which a multi-shot pyrolyzer (EGA/PY-3030D manufactured by Frontier Laboratories) was connected to a GC-QMS (JMS-Q1600GC UltraQuad™ SQ-Zeta, manufactured by JEOL Ltd.) was used.
The sample was heated from 50 ℃ to 1050 ℃ at a rate of 40 ℃ per minute in a heating furnace.
Generated gases including hydrogen are introduced into a fused silica capillary blank tube (length 2.5m, inner diameter
0.25mm) connected to the GC oven at a split ratio of 4:1. Those gases are detected directly by the mass spectrometer.
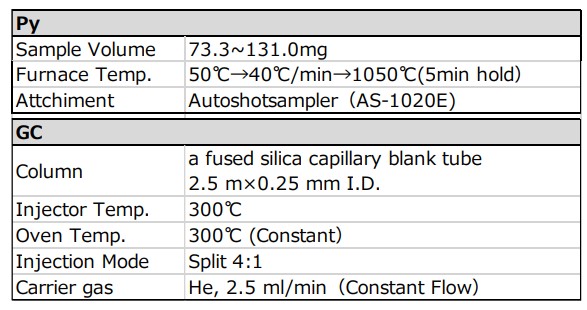
Table 1 shows the detailed conditions for pyrolysis GC/MS measurement.

JMS-Q1600GC UltraQuadTM SQ-Zeta
Table 1 Measurement condition

Result and Discussion
Figure 1 shows the direct-EGAMS measurement results of a steel pin (containing 7.9 ppm hydrogen). An extracted ion chromatogram (EIC) of m/z 2 tracing hydrogen is shown in the upper row, and an average mass spectrum for a retention time of 5 to 20 minutes is shown in the lower row. The horizontal scale of the EIC also shows the heating temperature of the sample as well as the holding time. Hydrogen profiles were confirmed in two temperature ranges from 250°C to 450°C and from 450°C to 850°C.
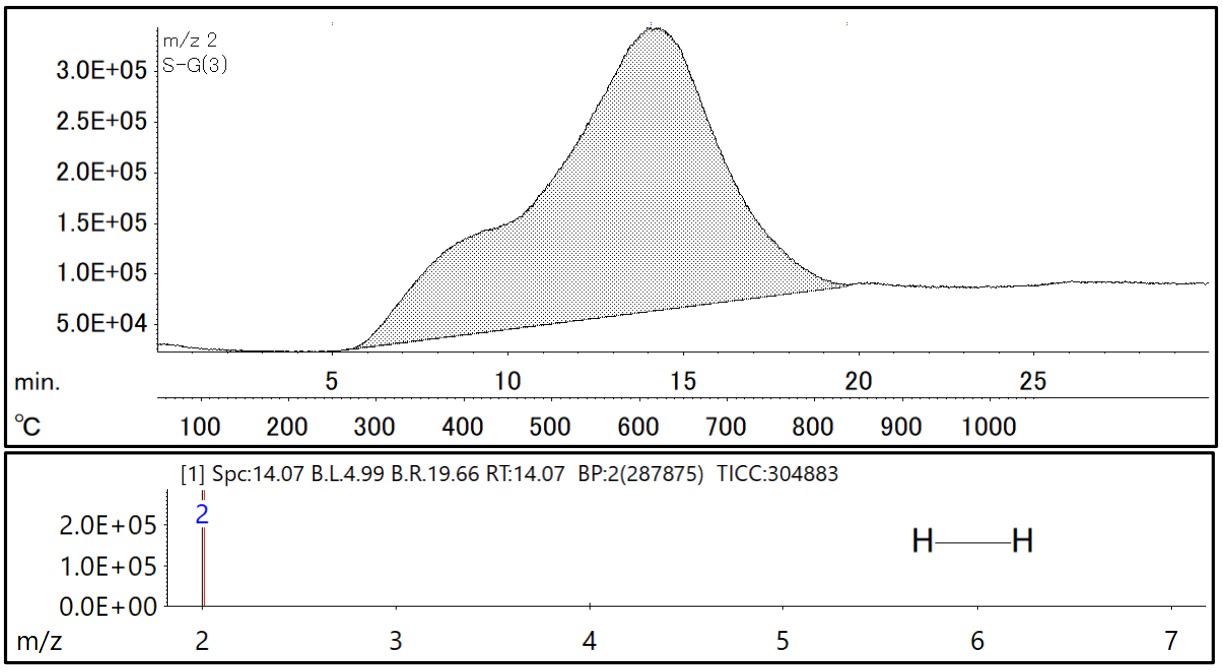
Figure 1 Extracted ion chromatogram(EIC) and Mass spectrum of Hydrogen
Figure 2 shows the result of re-measurement of the same sample after the measurement of the steel pin and the comparative EIC result of hydrogen detection in the measurement of two test pieces. In addition, Table 2 shows the sample weight and the obtained hydrogen peak area value in one and two steel test pieces, and the relative coefficient between the sample weight and the peak area value. Since no hydrogen peak was detected in the remeasurement of the steel pin, it is presumed that most of the hydrogen in the test piece is desorbed by one measurement. In addition, the amount of hydrogen detected was almost doubled with respect to the two test pins, indicating a good correlation between the weight of the sample and the amount of hydrogen detected.
Figure 3 shows the EIC results for three repeated measurements of steel specimens, and Table 2 shows the sample weight and the obtained hydrogen peak area value, and the relative coefficient between the sample weight and the peak area value. Good quantification was confirmed from the responsiveness and reproducibility of the hydrogen peak area by three consecutive measurements of the steel pin.
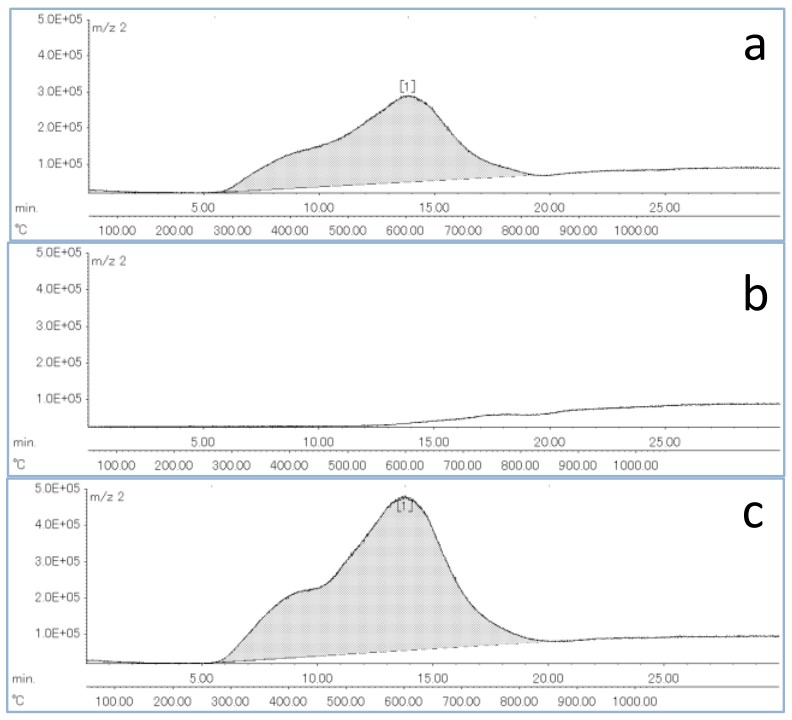
Figure 2 Extracted ion chromatograms of Hydrogen (a: one steel test piece, b: Remeasurement of the same sample, c: Two steel test pieces)
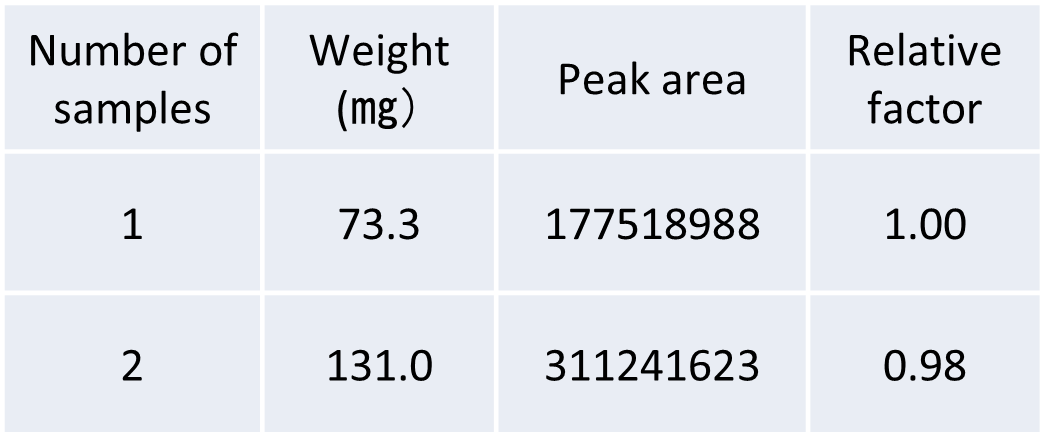
Table 2 Relationship between sample weight and peak area value for 1 and 2 steel test pieces
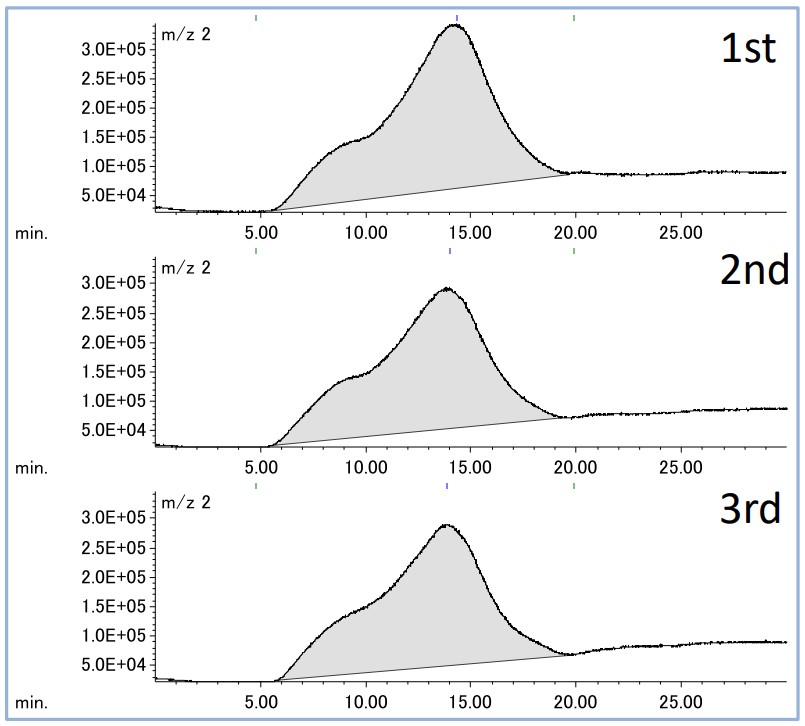
Figure 3 Extracted ion chromatograms for hydrogen repeated three times
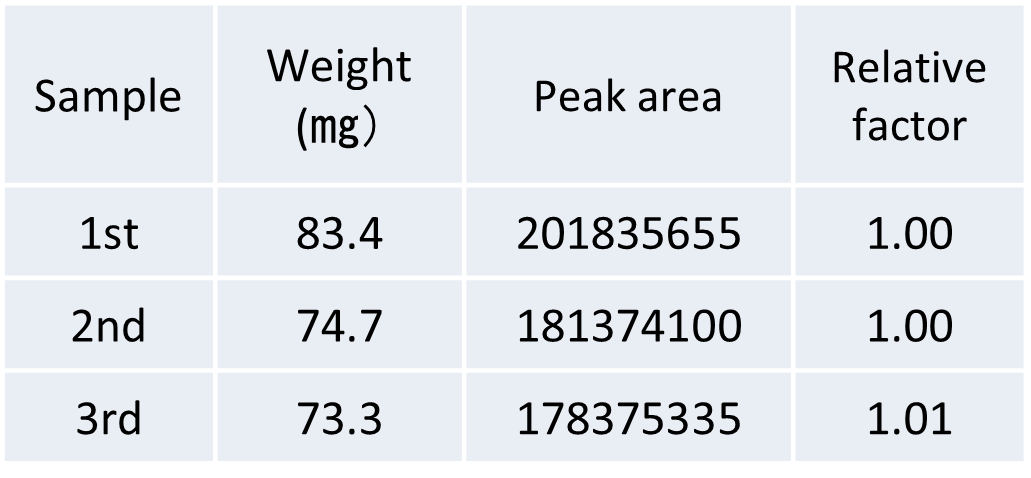
Table 3 Relationship between sample weight and peak area value in three repeated measurements of steel specimens
Conclusion
In this report, we reported an analysis example of hydrogen in steel pins using the direct-EGAMS method in which the hydrogen generated when the sample is heated is directly detected by a mass spectrometer.
From the results, it was confirmed that it is a qualitatively and quantitatively good analysis method for detecting hydrogen in steel pins.
Solutions by field
Related products
Are you a medical professional or personnel engaged in medical care?
No
Please be reminded that these pages are not intended to provide the general public with information about the products.

