Analyzing an Additive Component of Adhesive using Group Analysis Function of msFineAnalysis AI
MSTips No. 418
Introduction
Electron ionization (EI) is one of the most popular ionization methods used in gas chromatography-mass spectrometry (GC-MS). Consequently, compounds are typically identified by a mass spectral database search using EI mass spectra. Because molecular ions are often weak or absent in 70 eV EI mass spectra, identification of unknowns can be difficult by EI alone. In these cases, soft ionization (SI) can be very helpful for producing and identifying molecular ions. Recently, JEOL began developing an integrated qualitative analysis workflow that automatically combines and interprets the information from EI and SI data1). And then in 2018, we introduced our integrated qualitative analysis software "msFineAnalysis" which uses both EI and SI data to improve compound identification for GC-MS applications. Despite the fact that msFineAnalysis was automatically able to determine the molecular formula and partial structure information from EI fragment ion formulas, the actual structural formulas still required manual analysis using chemical compositions. To address this, we then developed an automated structure analysis software package entitled "msFineAnalysis AI" which uses artificial intelligence (AI) to predict EI mass spectra from chemical structures2).
The msFineAnalysis AI software has a Group analysis function that enables easy extraction of specific compounds. In MSTips No. 417, we introduced an overview of group analysis function and example of its application for vinyl acetate resin samples. The group analysis function includes a list of characteristic fragment ions and neutral losses as described in MSTips No. 417, as well as an "additive list" containing major additives for polymeric materials and an "off-flavor list" containing off-flavor components. In this MSTips, we will introduce additive compounds analysis results using this group analysis function with "additive list".
Experimental

msFineAnalysis AI
A commercially-available adhesive was used as a test sample in this study. We performed Py-GC-HRTOFMS measurements using both EI and field ionization (FI) modes with a combination EI/FI ion source. The qualitative data processing was performed with msFineAnalysis AI (JEOL).
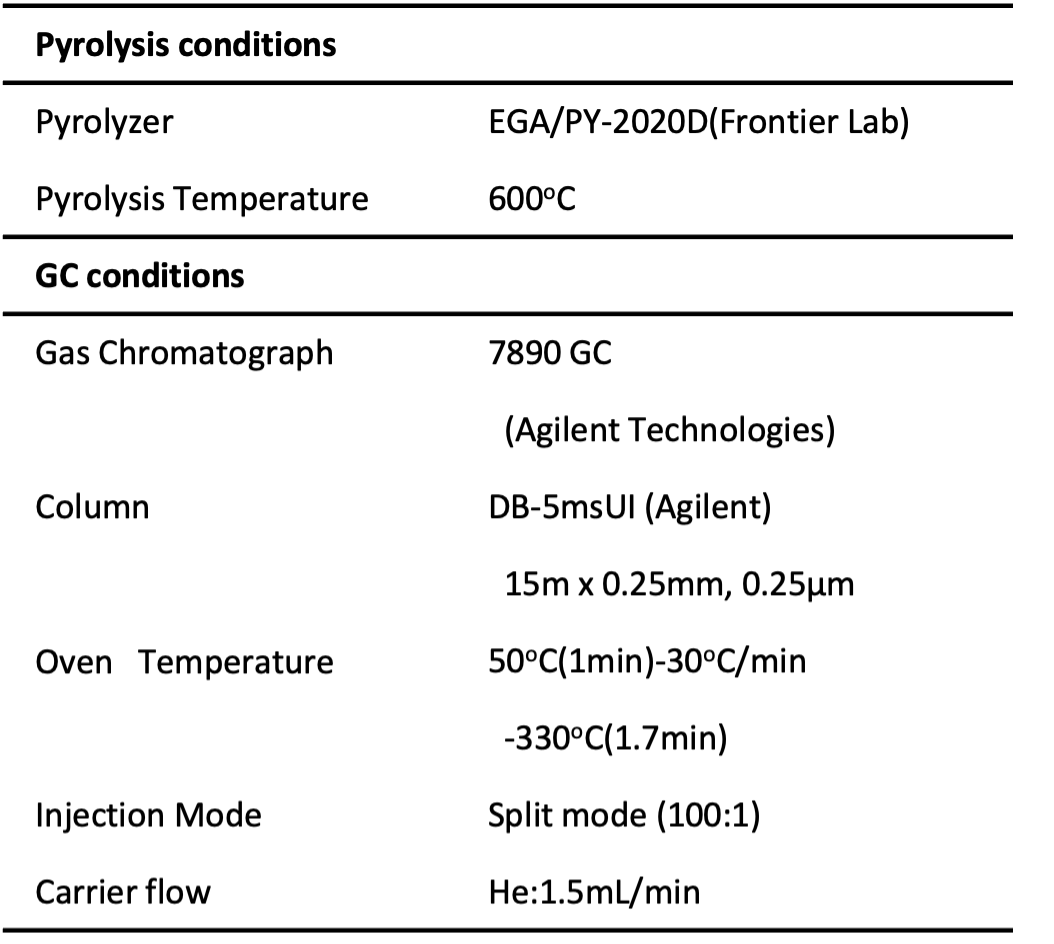
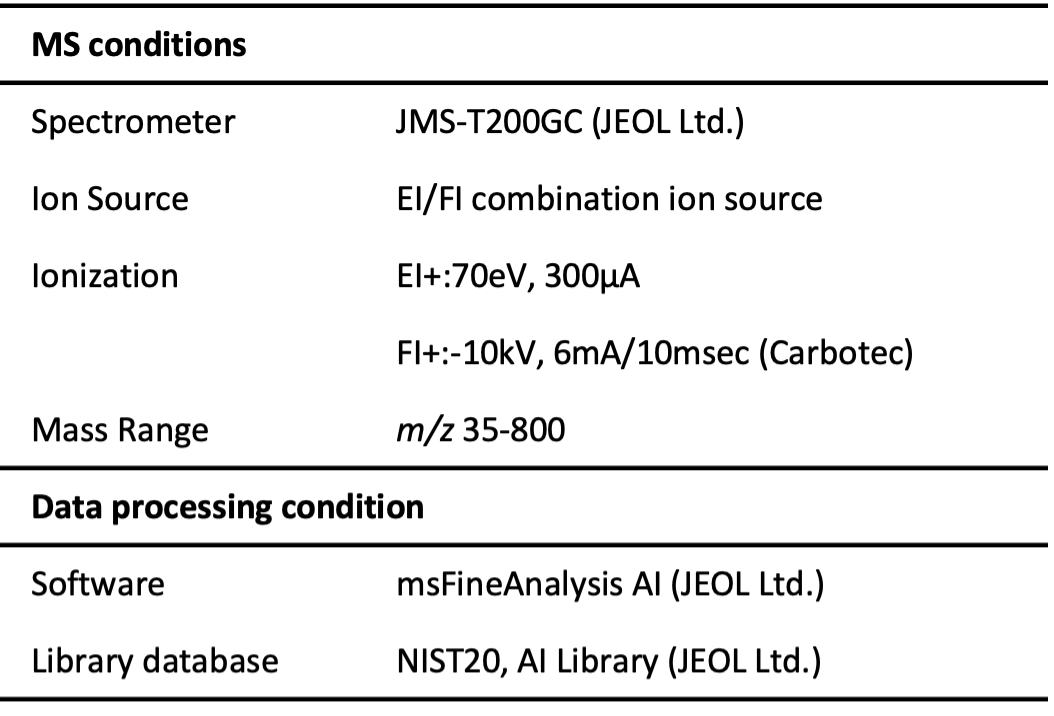
Measurement conditions are shown in Table 1.
Table 1 Measurement and analysis conditions
Results and Discussion
Extraction of additive component by group analysis
Figure 1 shows an example of additive analysis result using group analysis function with "additive list".
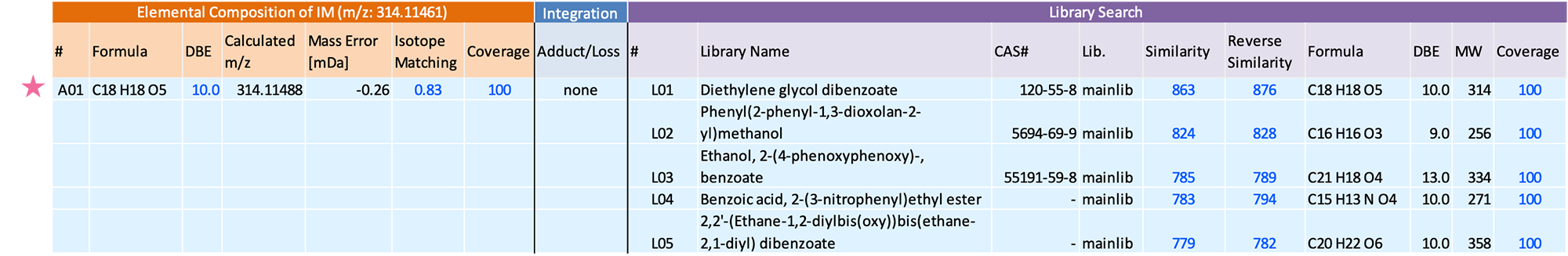
The chromatographic view on the left top shows the GC/EI data with the TICC marked by a solid black line. The bottom left view shows the soft ionization data with the TICC marked by a solid green line. The view on the right shows "Ion List" detected in the analysis data. The additive list contains molecular formulas of 525 additives for polymeric materials. If a compositional formula registered in this additive list is present in the analysis data, the additive type and CAS number will be displayed in the Description column. In this study, a plasticizer with molecular formula C18H18O5 (CAS No. 120-55-8) was confirmed.
The blue peaks in each data of Figure1 represent the components containing C18H18O5+ extracted from the chromatographic deconvolution result. The operator can select an ion such as C18H18O5+ from the table and click the OK button at the bottom right of the GUI to immediately create a C18H18O5 tab, thus allowing for extraction of the components containing C18H18O5+ (Figure 2).
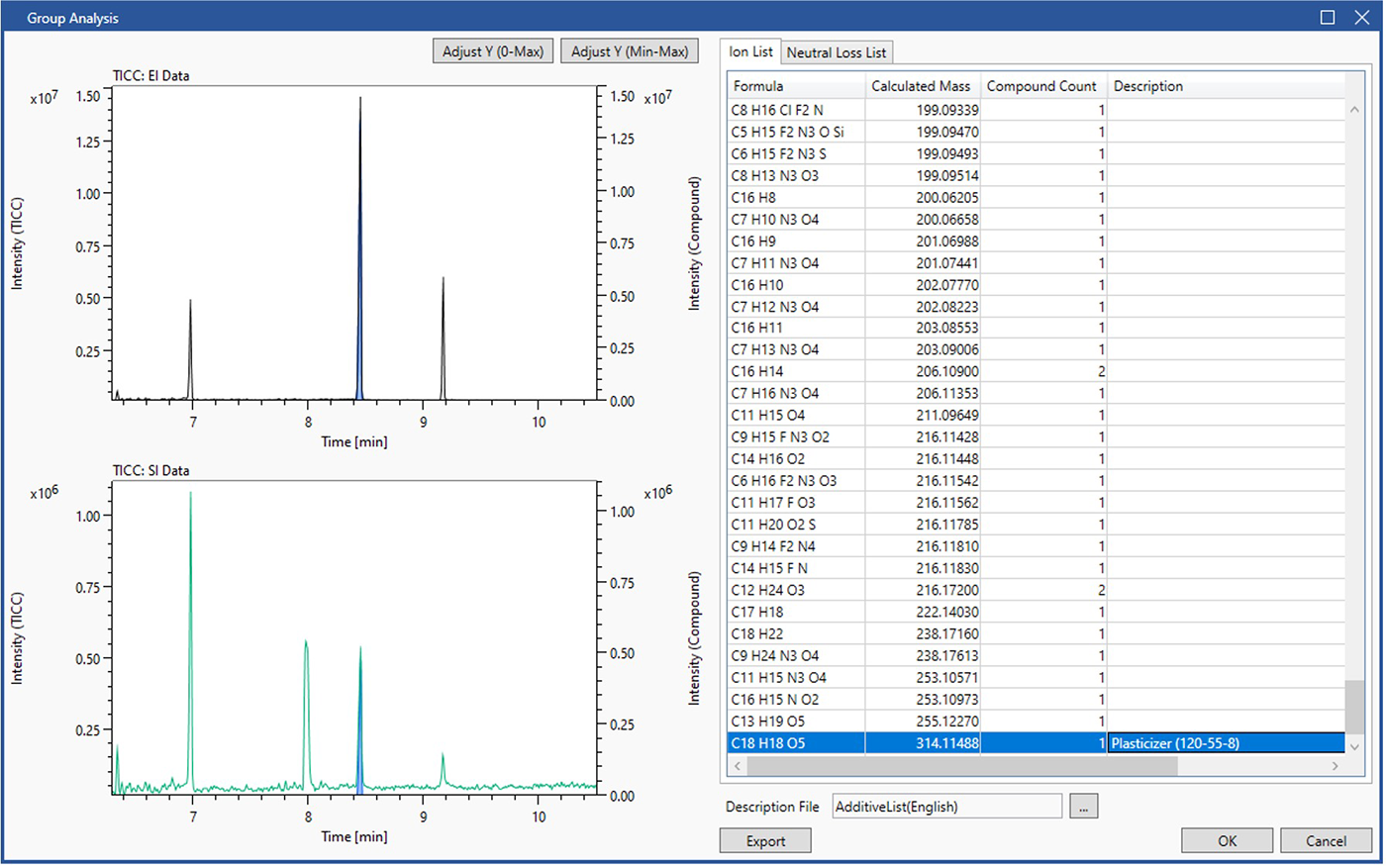
Figure 1 Group analysis result using additive list
Figure 2 shows the extracted results for component containing C18H18O5+ . The Group Analysis function displays an "All" tab for the entire analysis results and up to 5 tabs for groups created for ions or neutral losses specified from the exact mass list in Figure 1. The ID and integrated analysis results are then shared between tabs. A total of 60 component peaks were observed in the analysis results, but it was possible to immediately extract only the peak containing C18H18O5+. Detailed qualitative analysis results for this component are described in the next section.
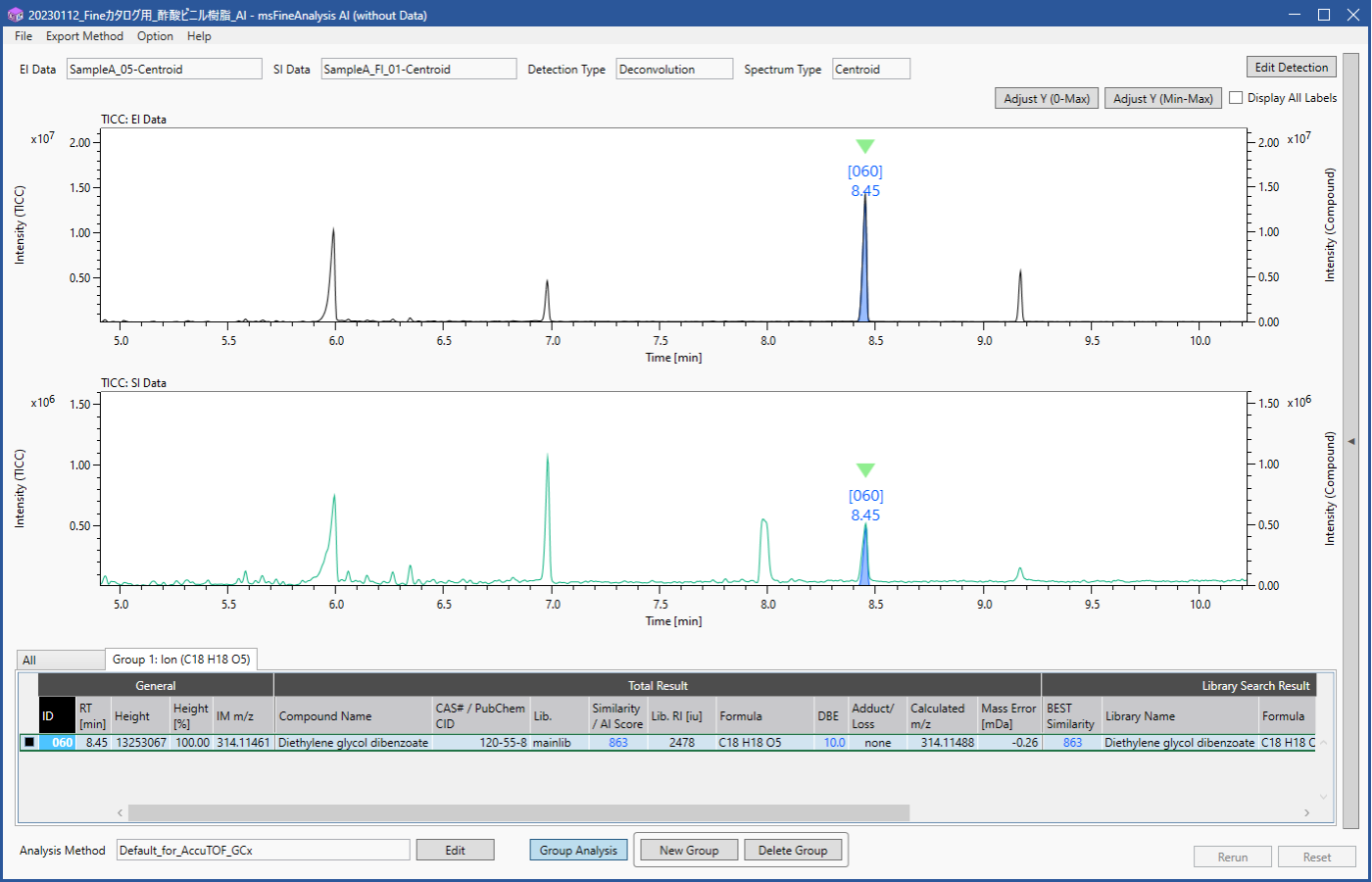
Figure 2 Group analysis result for C18H18O5 ion
Qualitative analysis of additive component
Figure 3 shows the mass spectra of the component extracted by group analysis function. m/z 314, which is presumed to be the molecular ion, was detected only in the FI, while no molecular ion was detected in the EI. The integrated qualitative analysis result list (top 5 candidates) by msFineAnalysis AI is shown in Table 2. In the NIST Library DB search result, several highly similar compounds were presented. However, based on the estimated elemental composition of the molecular ion, the molecular formula was calculated to be C18H18O5. Therefore, this component was presumed to be "Diethylene glycol dibenzoate" (Figure 4). From the above results, it was confirmed that additive component was detected from the commercially available adhesive measured this time.
Even in an analysis such as pyrolysis GC-MS analysis in which many peaks are detected, additive-derived peak could be extracted immediately.
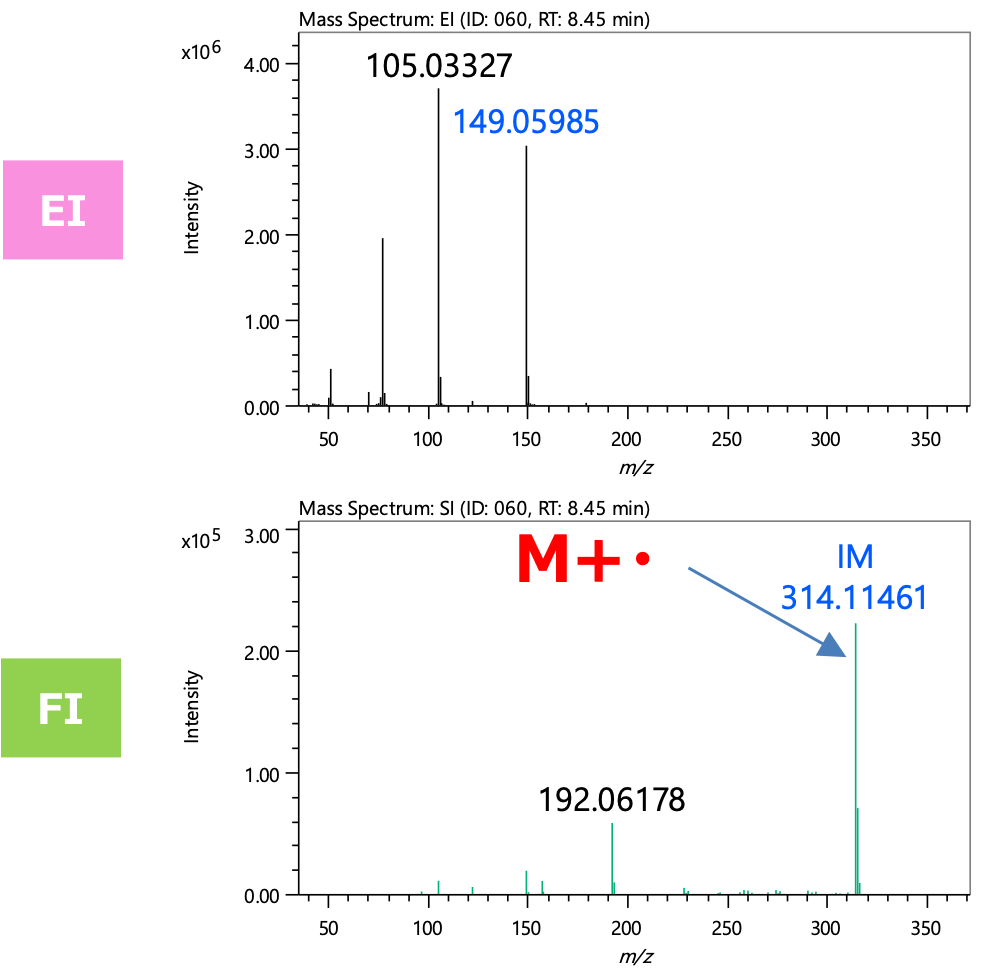
Figure 3 Mass spectra
Figure 4 Structural formula of Diethylene
glycol dibenzoate
Table 2 Integrated qualitative analysis result using the msFineAnalysis AI
Conclusion
In this MSTips, we introduced an example of extracting additive component in non-target analysis using the group analysis function of msFineAnalysis AI. By using the group analysis function, target analysis-like analysis can be performed even in non-target analysis, so detailed analysis in a shorter time can be expected.
Reference
- M. Ubukata, A. Kubo, K. Nagatomo, T. Hizume, H. Ishioka, A. J. Dane, R. B. Cody, Y. Ueda. Integrated qualitative analysis of polymer sample by pyrolysis–gas chromatography combined with high-resolution mass spectrometry: Using accurate mass measurement results from both electron ionization and soft ionization. Rapid Commun Mass Spectrom. 2020; 34:e8820.
- A. Kubo , A. Kubota, H. Ishioka, T. Hizume, M. Ubukata, K. Nagatomo, T. Satoh, M. Yoshida, F. Uematsu. Construction of a mass spectrum library containing predicted electron ionization mass spectra prepared using a machine learning model and the development of an efficient search method. Mass Spectrometry. 2023; 12: A0120.
Solutions by field
Related products
Are you a medical professional or personnel engaged in medical care?
No
Please be reminded that these pages are not intended to provide the general public with information about the products.


