Chromatographic methods of quantification and NMR quantification
Issue 2
Measured with NMR
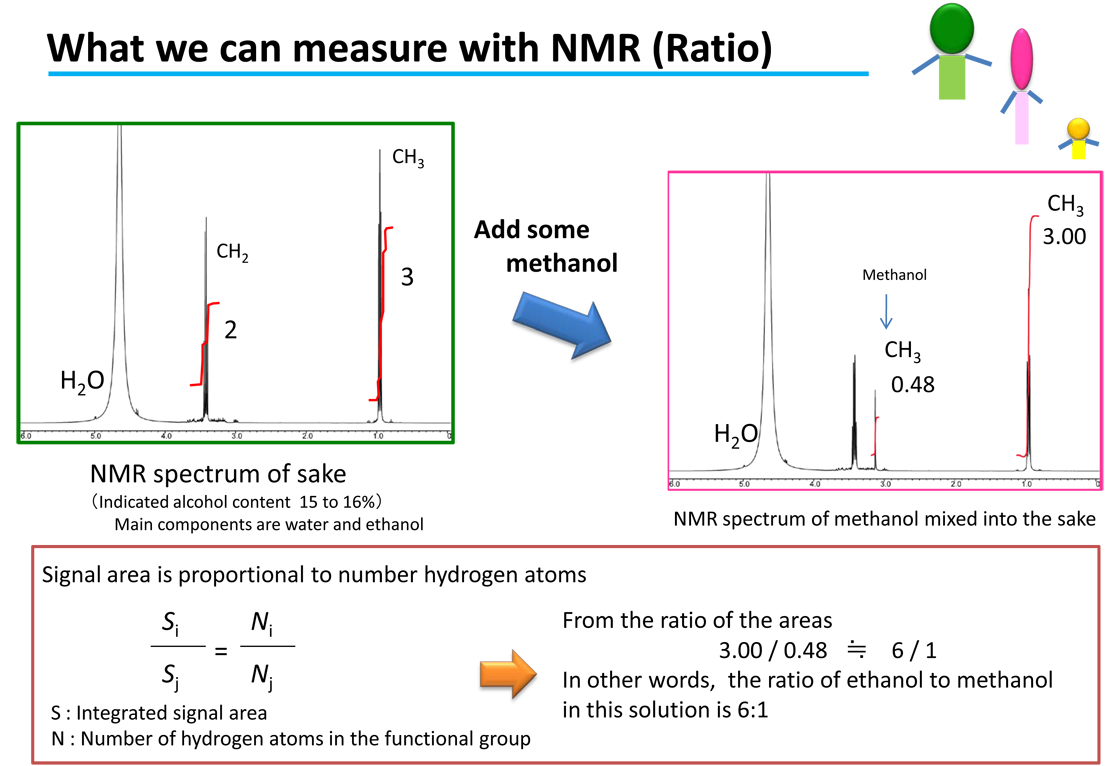
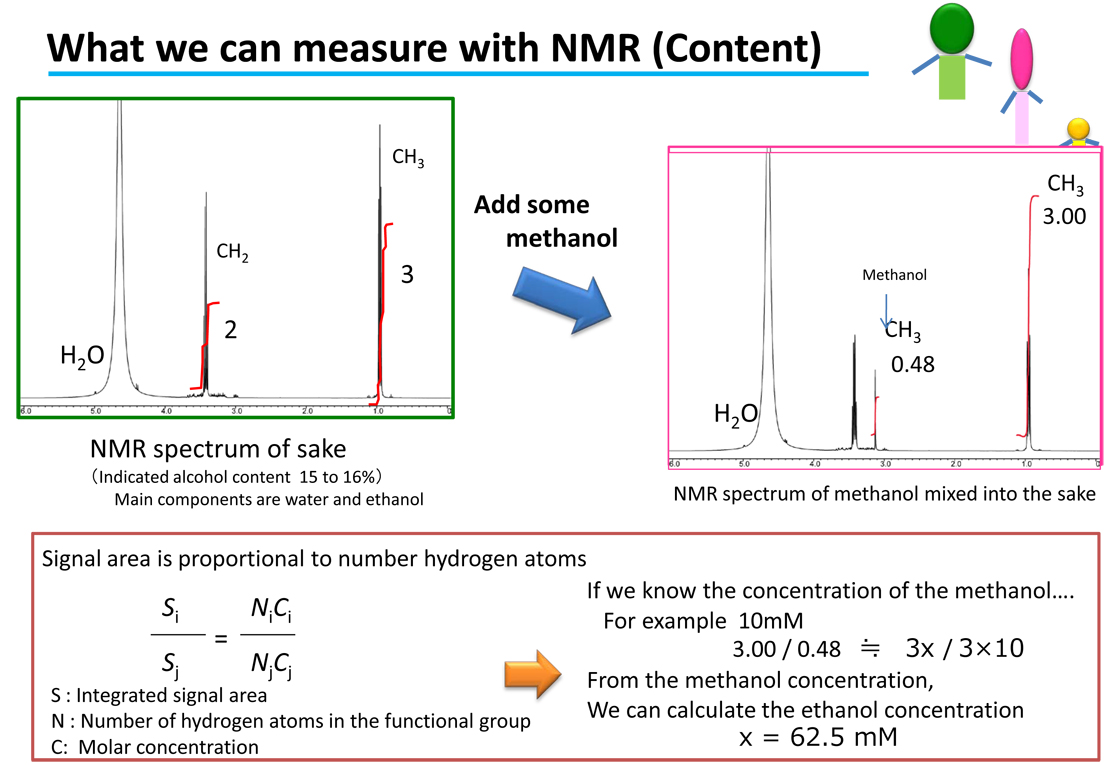
In the previous issue it was explained how the areas of the signals on the NMR spectrum are used to determine the ratios of components in the sample. This is a reflection of the principle of NMR, which is that the areas of the signals are proportional to the number of hydrogen atoms (number of protons). Since the nuclear spins are observed directly, in terms of molar concentration, these relationships also hold intermolecule . In other words, if we know the molar concentration of one of the substances providing a signal, it can be used as a reference to determine the molar concentrations of the other components. For example, if we know that the molar concentration of the methanol is 10 mM, we can use this to calculate that the ethanol is 62.5 mM. Thus, “what we can learn with NMR” is not just the ratio between various components, it is also possible to determine the amount of a target component.
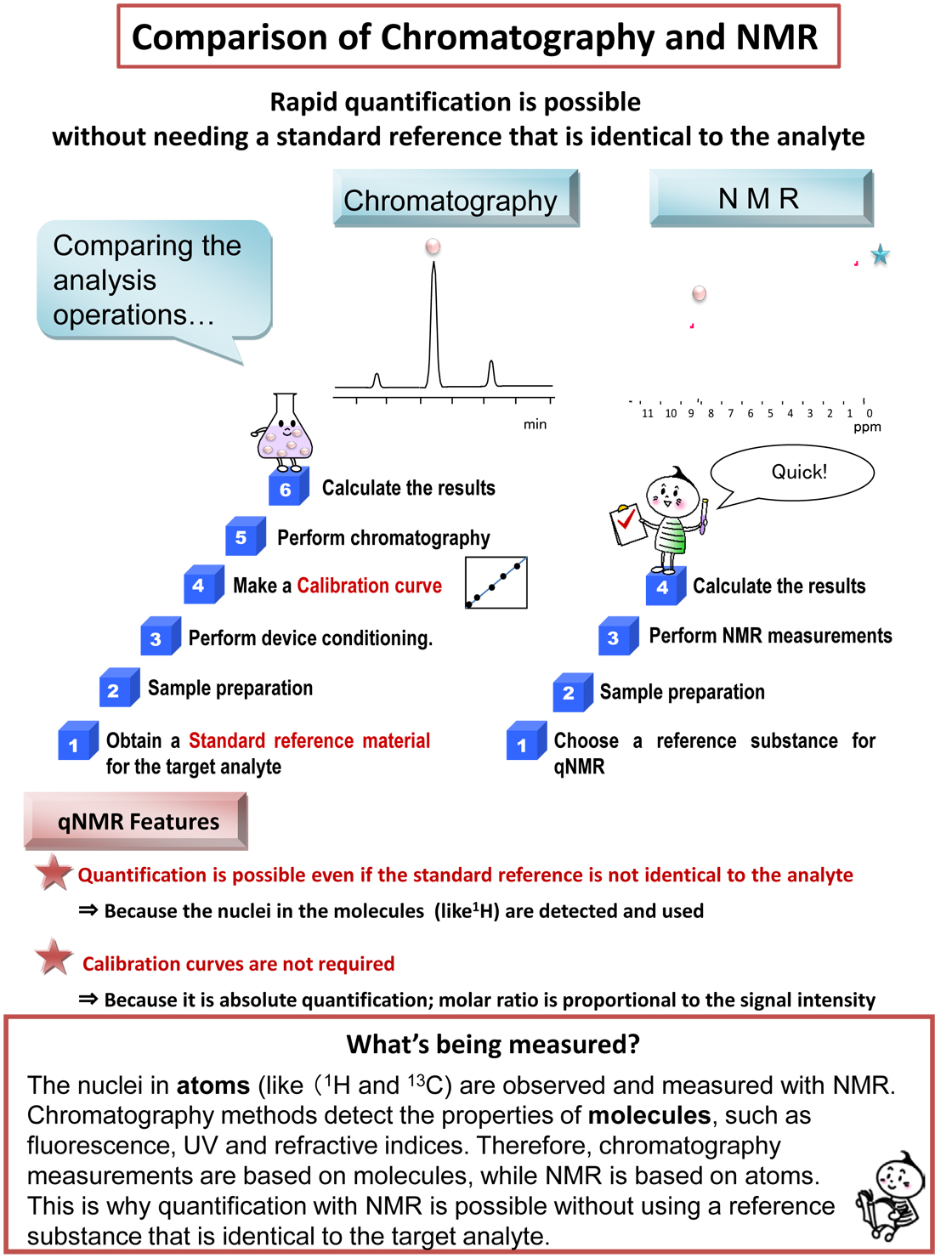
Chromatography vs. NMR quantification
An example of the quantification (measurement of the amount) of ethanol from the methanol content was shown in [What we can measure with NMR]. Some readers probably will already have noticed that quantitative analysis with NMR can be performed even if the quantitative standard reference sample is not the same compounds as the analyte. This is because the hydrogen (protons 1H) within the molecules is observed and measured with NMR. Even if the molecules are different, as long as there is hydrogen present in the molecule, quantitative analysis is possible. Accordingly, there is no need for calibration curves for quantitative analysis with NMR. That means that we can perform quantitative analysis even when we do not have standard reference samples for the sample being analyzed (such as a new synthetic compound or extract). Attachment (Figure: Comparison of analysis operations)
Are you a medical professional or personnel engaged in medical care?
No
Please be reminded that these pages are not intended to provide the general public with information about the products.
